To minimize contact with oxygen and water, alkali metals must be stored in an airtight container under mineral oil and/or under an inert gas, such as argon. Do not work alone when handling alkali metals.Proper street clothing – long pants (or equivalent clothing that covers the legs and ankles) and closetoed, non-perforated shoes that completely cover the feet.Safety glasses o Gloves of the appropriate chemical resistance.Minimum PPE for alkali metal work includes: Resources for airsensitive techniques include Sigma Aldrich Technical Bulletin AL-134 and ChemistryViews Tips and Tricks for the Lab: Air-Sensitive Techniques. O Use air-sensitive techniques when handling alkali metals outside of a glove box. O To prevent reaction with air, keep alkali metals under an inert solvent such as mineral oil, hexanes, or toluene. The work area must be free of water, extraneous flammable materials, and sparks or other ignition sources. If it is necessary to work with these metals outside of a glove box, perform this work in a certified laboratory fume hood.When possible, work with alkali metals in a glove box, as a glove box can provide an inert working environment.ABC and CO2 fire extinguishers must not be used to extinguish alkali metal fires. Contact the Stanford University Fire Marshal’s Office at (650) 723-0448 to obtain these extinguishers. Ensure that an appropriate Class D fire extinguisher is available prior to beginning work with alkali metals.Ensure personnel working with alkali metals have completed appropriate Tier III (laboratory-specific) training, which includes a review of the SOP.Develop a Standard Operating Procedure (SOP) for your experiment and review the SOP with your Principal Investigator (PI).The heavier alkali metals will react more violently with water. The heat produced by this reaction may ignite the hydrogen or the metal itself, resulting in a fire or an explosion. 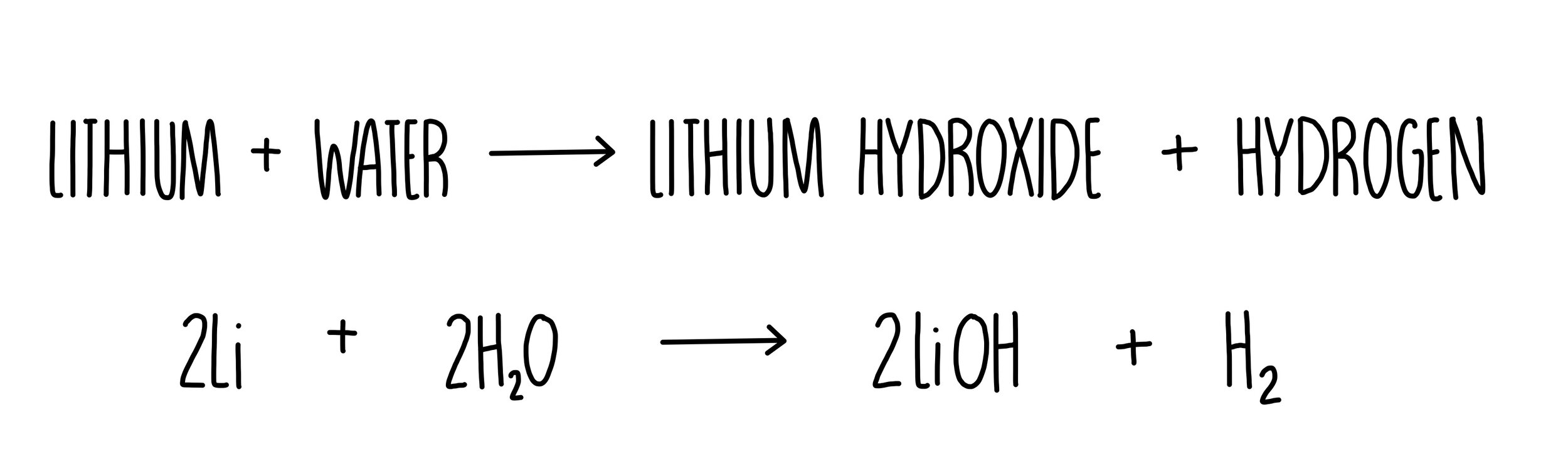
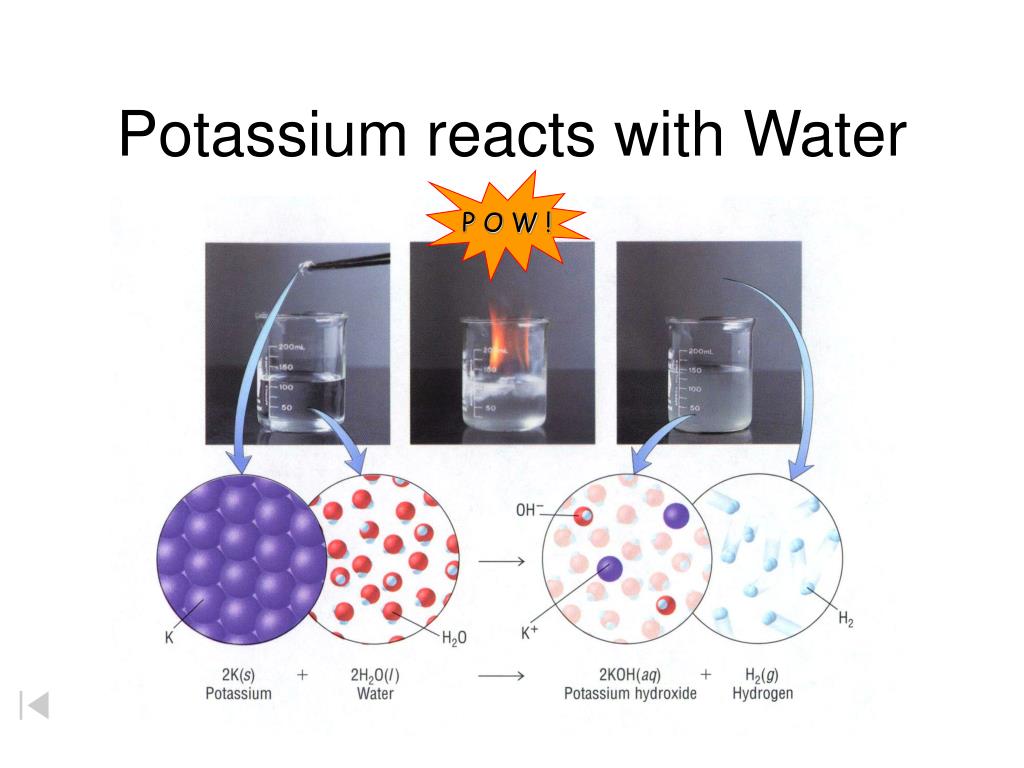
The heavier alkali metals (rubidium and cesium) will spontaneously ignite upon exposure to air at room temperature.Īlkali metals react with water to produce heat, hydrogen gas, and the corresponding metal hydroxide. What are the hazards?Īlkali metals react with air to form caustic metal oxides. Lithium, sodium, potassium, rubidium, and cesium (L-R). Due to their reactivity, special precautions must be taken when using and storing these metals. Alkali metals react readily with air and moisture. In their pure forms, the alkali metals (lithium, sodium, potassium, rubidium, and cesium) are soft, shiny metals with low melting points.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
